Propylene glycol
 | |||
| |||
| Names | |||
|---|---|---|---|
Preferred IUPAC name Propane-1,2-diol | |||
| Other names Propylene glycol α-Propylene glycol 1,2-Propanediol 1,2-Dihydroxypropane Methyl ethyl glycol (MEG) Methylethylene glycol | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
ChEBI |
| ||
ChEMBL |
| ||
ChemSpider |
| ||
ECHA InfoCard | 100.000.307 | ||
EC Number | 200-338-0 | ||
E number | E1520 (additional chemicals) | ||
PubChem CID |
| ||
RTECS number | TY6300000 | ||
UNII |
| ||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula | C3H8O2 | ||
Molar mass | 76.10 g·mol−1 | ||
Density | 1.036 g/cm3 | ||
Melting point | −59 °C (−74 °F; 214 K) | ||
Boiling point | 188.2 °C (370.8 °F; 461.3 K) | ||
Solubility in water | Miscible | ||
Solubility in ethanol | Miscible | ||
Solubility in diethyl ether | Miscible | ||
Solubility in acetone | Miscible | ||
Solubility in chloroform | Miscible | ||
log P | -1.34[2] | ||
Thermal conductivity | 0.34 W/m-K (50% H2O @ 90 °C (194 °F)) | ||
Viscosity | 0.042 Pa·s | ||
| Pharmacology | |||
ATCvet code | QA16QA01 (WHO) | ||
| Hazards | |||
S-phrases (outdated) | S24 S25 | ||
NFPA 704 |  1 0 0 | ||
| Related compounds | |||
Related glycols | Ethylene glycol, 1,3-propanediol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
Infobox references | |||
Propylene glycol (IUPAC name: propane-1,2-diol) is a synthetic organic compound with the chemical formula C3H8O2. It is a viscous, colorless liquid which is nearly odorless but possesses a faintly sweet taste. Chemically it is classed as a diol and is miscible with a broad range of solvents, including water, acetone, and chloroform.
It is produced on a large scale and is primarily used in the production of polymers, but also sees use in food processing, and as a process fluid in low-temperature heat-exchange applications. In the European Union, it has the E-number E1520 for food applications. For cosmetics and pharmacology, the number is E490. Propylene glycol is also present in propylene glycol alginate which known as E405.
The compound is sometimes called (alpha) α-propylene glycol to distinguish it from the isomer propane-1,3-diol, known as (beta) β-propylene glycol.
Contents
1 Structure and properties
2 Production
2.1 Industrial
2.2 Laboratory
3 Applications
4 Safety in humans
4.1 Oral administration
4.2 Skin, eye and inhalation contact
4.3 Intravenous administration
4.4 Animals
4.5 Allergic reaction
5 Environmental
6 References
7 External links
Structure and properties
Propylene glycol is a clear, colorless and hygroscopic liquid. Propylene glycol contains an asymmetrical carbon atom, so it exists in two enantiomers. The commercial product is a racemic mixture. Pure optical isomers can be obtained by hydration of optically pure propylene oxide.[4]
The freezing point of water is depressed when mixed with propylene glycol, owing to the effects of dissolution of a solute in a solvent (freezing-point depression). In general, glycols are non-corrosive, have very low volatility and very low toxicity; however, the closely related ethylene glycol (a key ingredient in antifreeze) is toxic to humans and to many animals.[5]
Production
Industrial
Industrially, propylene glycol is produced from propylene oxide[6] (for food-grade use), and global capacity in 1990 was 900,000 tonnes per year.[7] Different manufacturers use either non-catalytic high-temperature process at 200 °C (392 °F) to 220 °C (428 °F), or a catalytic method, which proceeds at 150 °C (302 °F) to 180 °C (356 °F) in the presence of ion exchange resin or a small amount of sulfuric acid or alkali.

Final products contain 20% propylene glycol, 1.5% of dipropylene glycol and small amounts of other polypropylene glycols.[8] Further purification produces finished industrial grade or USP/JP/EP/BP grade propylene glycol that is typically 99.5% or greater. Propylene glycol can also be converted from glycerol, a biodiesel byproduct. This starting material is usually reserved for industrial use because of the noticeable odor and taste that accompanies the final product.
Laboratory
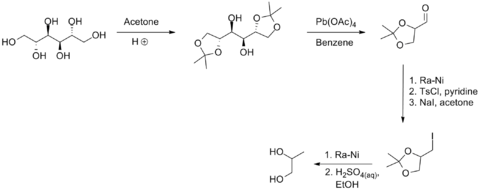
S-Propanediol may be synthesized from D-mannitol, through the following scheme:[9]
Applications

Propylene glycol is often used in electronic cigarettes.

A bottle of flavored "e-liquid" for "vaping" shows propylene glycol as the main ingredient

Propylene glycol is commonly used to de-ice aircraft
Forty-five percent of propylene glycol produced is used as chemical feedstock for the production of unsaturated polyester resins. In this regard, propylene glycol reacts with a mixture of unsaturated maleic anhydride and isophthalic acid to give a copolymer. This partially unsaturated polymer undergoes further crosslinking to yield thermoset plastics. Related to this application, propylene glycol reacts with propylene oxide to give oligomers and polymers that are used to produce polyurethanes.[7] Propylene glycol is used in waterbased acrylic architectural paints to extend dry time which it accomplishes by preventing the surface from drying due to its slower evaporation rate compared to water. Propylene glycol is used as a humectant (E1520), solvent,[10] and preservative in food and for tobacco products. It is also one of the major ingredients (<1–92%), along with Vegetable Glycerin (VG), of the e-liquid and cartridges used in electronic cigarettes where it is aerosolized in the atomizer.[11] It is also used in model railway and model boat applications to simulate steam or smoke, using a small heating element.
Propylene glycol is also used in various edible items such as coffee-based drinks, liquid sweeteners, ice cream, whipped dairy products and soda.[12][13]Vaporizers used for delivery of pharmaceuticals or personal-care products often include propylene glycol among the ingredients.[7] In alcohol-based hand sanitizers, it is used as a humectant to prevent the skin from drying.[14] Propylene glycol is used as a solvent in many pharmaceuticals, including oral, injectable and topical formulations, such as for diazepam and lorazepam which are insoluble in water.[15] Certain formulations of artificial tears, such as Systane, use proplyene glycol as an ingredient.[16]
Like ethylene glycol, propylene glycol is able to lower the freezing point of water, and so it is used as aircraft de-icing fluid.[7][17] Water-propylene glycol mixtures dyed pink to indicate the mixture is relatively nontoxic are sold under the name of RV or marine antifreeze. Propylene glycol is frequently used as a substitute for ethylene glycol in low toxicity, environmentally friendly automotive antifreeze. It is also used to winterize the plumbing systems in vacant structures.[18] The eutectic composition/temperature is 60:40 propylene glycol:water/-60 °C.[19][20] The −50 °F/−45 °C commercial product is, however, water rich; a typical formulation is 40:60.[21]
Propylene glycol is used in veterinary medicine as an oral treatment for hyperketonaemia in ruminants. Glucose, which can be used in non-ruminants for this purpose, is not effective due to its consumption by the resident microbes of the rumen. Propylene glycol is partially metabolized in the rumen to propionate which can be used as an energy source. The remainder is absorbed into the bloodstream and used by the liver for gluconeogenesis.[22]
Safety in humans
Oral administration
The acute oral toxicity of propylene glycol (E1520) is very low, and large quantities are required to cause perceptible health damage in humans; propylene glycol is metabolized in the human body into pyruvic acid (a normal part of the glucose-metabolism process, readily converted to energy), acetic acid (handled by ethanol-metabolism), lactic acid (a normal acid generally abundant during digestion),[23] and propionaldehyde (a potentially hazardous substance).[24][25][26] According to the Dow Chemical Company, The LD50[27] (Lethal Dose that kills in 50% of tests,) for rats is [28] 20 g/kg (rat/oral).
Serious toxicity generally occurs at plasma concentrations over 4 g/L, which requires extremely high intake over a relatively short period of time, or when used as a vehicle for drugs or vitamins given intravenously or orally.[29] It would be nearly impossible to reach toxic levels by consuming foods or supplements, which contain at most 1 g/kg of PG, except for alcoholic beverages in the US which are allowed 5 percent = 50g/kg.[30] Cases of propylene glycol poisoning are usually related to either inappropriate intravenous administration or accidental ingestion of large quantities by children.[31]
The potential for long-term oral toxicity is also low. In one study, in 1972, 12 rats were provided with feed containing as much as 5% PG over a period of 2 years, and showed no apparent ill effects; no data on offspring were offered.[32] Because of its low chronic oral toxicity, propylene glycol was classified by the U. S. Food and Drug Administration as "generally recognized as safe" (GRAS) for use as a direct food additive, including frozen foods such as ice cream and frozen desserts.[30][33] The GRAS designation is specific to its use in food, and does not apply to other uses.[34]
Skin, eye and inhalation contact
Prolonged contact with propylene glycol (E490) is essentially non-irritating to the skin.[35] Undiluted propylene glycol is minimally irritating to the eye, producing slight transient conjunctivitis; the eye recovers after the exposure is removed. Exposure to mists may cause eye irritation, as well as upper respiratory tract irritation. Inhalation of propylene glycol vapors appears to present no significant hazard in ordinary applications.[36] However, limited human experience indicates that inhalation of propylene glycol mists could be irritating to some individuals.[37] It is therefore recommended that propylene glycol not be used in applications where inhalation exposure or human eye contact with the spray mists of these materials is likely, such as simulated fogs for theatrical productions, or antifreeze solutions for emergency eye wash stations.[38]
This is of particular concern however, due to the relatively new trend of "vaping", also known as use of e-cigarettes, due to many of the commercially available "juice" products containing propylene glycol as a key ingredient, along with varying concentrations of nicotine & other chemicals, and it being repetitively inhaled by e-cigarette users.
According to a 2010 study by Karlstad University, the concentrations of PGEs (counted as the sum of propylene glycol and glycol ethers) in indoor air, particularly bedroom air, has been linked to increased risk of developing numerous respiratory and immune disorders in children, including asthma, hay fever, eczema, and allergies, with increased risk ranging from 50% to 180%. This concentration has been linked to use of water-based paints and water-based system cleansers. However, the study authors write that glycol ethers and not propylene glycol are the likely culprit.[39][40][41]
Propylene glycol does not cause sensitization, and it shows no evidence of being a carcinogen or of being genotoxic.[42][43]
Intravenous administration
Adverse responses to intravenous administration of drugs that use propylene glycol as an excipient have been seen in a number of people, particularly with large dosages. Responses may include "hypotension, bradycardia... QRS and T abnormalities on the ECG, arrhythmia, cardiac arrest, serum hyperosmolality, lactic acidosis, and haemolysis".[44] A high percentage (12% to 42%) of directly-injected propylene glycol is eliminated or secreted in urine unaltered depending on dosage, with the remainder appearing in its glucuronide-form. The speed of renal filtration decreases as dosage increases,[45] which may be due to propylene glycol's mild anesthetic / CNS-depressant -properties as an alcohol.[46] In one case, intravenous administration of propylene glycol-suspended nitroglycerin to an elderly man may have induced coma and acidosis.[47]
Animals
Propylene glycol is an approved food additive for dog and sugar glider food under the category of animal feed and is generally recognized as safe for dogs,[48] with an LD50 of 9 mL/kg. The LD50 is higher for most laboratory animals (20 mL/kg).[49] However, it is prohibited for use in food for cats due to links to Heinz body anemia.[50]
Allergic reaction
Individuals who cannot tolerate propylene glycol experience inflamed dry skin in the facial area, or small red dots on the body. Investigators believe that the incidence of allergic contact dermatitis to propylene glycol may be greater than 2% in patients with eczema or fungal infections, which are very common in countries with lesser sun exposure and lower-than-normal vitamin D balances. Therefore, propylene glycol allergy is more common in those countries.[51]
Environmental
Propylene glycol is known to exert high levels of biochemical oxygen demand (BOD) during degradation in surface waters. This process can adversely affect aquatic life by consuming oxygen needed by aquatic organisms for survival. Large quantities of dissolved oxygen (DO) in the water column are consumed when microbial populations decompose propylene glycol.[52]:2–23
Sufficient dissolved oxygen levels in surface waters are critical for the survival of fish, macroinvertebrates, and other aquatic organisms. If oxygen concentrations drop below a minimum level, organisms emigrate, if able and possible, to areas with higher oxygen levels or eventually die. This effect can drastically reduce the amount of usable aquatic habitat. Reductions in DO levels can reduce or eliminate bottom feeder populations, create conditions that favor a change in a community’s species profile, or alter critical food-web interactions.[52]:2–30
References
^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals. Merck & Co. ISBN 091191028X..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ "Propylene Glycol_msds".
^ "PROPYLENE GLYCOL - CAMEO Chemicals". NOAA Office of Response and Restoration. NOAA. Retrieved 3 October 2018.
^ "1,2-Propanediol". ChemIndustry.ru. Retrieved 2007-12-28.
^ "Ethylene Glycol and Propylene Glycol Toxicity What Are the Stages of Ethylene Glycol Intoxication?". Agency for Toxic Substances and Disease Registry. October 3, 2007.
^ Chauvel, Alain; Lefebvre, Gilles. Petrochemical Processes. Volume 2: Major Oxygenated, Chlorinated and Nitrated Derivatives. Editions Technip. p. 26. ISBN 9782710805632.
^ abcd Ullmann's Encyclopedia of Industrial Chemistry. John Wiley and Sons, Inc. ISBN 9783527306732.
^ "1,2-propanediol: chemical product info at CHEMINDUSTRY.RU". Retrieved 3 October 2018.
^ Hanessian, Stephen (1983). Total Synthesis of Natural Products: The 'Chiron' Approach. Pergamon press. p. 41. ISBN 978-0080307152.
^ Bradley, Jean-Claude; Abraham, Michael H; Acree, William E; Lang, Andrew (2015). "Predicting Abraham model solvent coefficients". Chemistry Central Journal. 9 (1): 12. doi:10.1186/s13065-015-0085-4. ISSN 1752-153X.
^ Simon, Harvey. "Electronic cigarettes: Help or hazard?". Harvard Medical School - Harvard Health Publications. Retrieved Sep 22, 2011.
^ "Quackmail: Why You Shouldn't Fall For The Internet's Newest Fool, The Food Babe". Butterworth, Trevor. Forbes. 16 June 2014. Retrieved 18 March 2015.
^ G. Jackson, R. T. Roberts and T. Wainwright (January 1980). "Mechanism of Beer Foam Stabilization by Propylene Glycol Alginate". Journal of the Institute of Brewing. 86 (1): 34–37. doi:10.1002/j.2050-0416.1980.tb03953.x.
^ Lohrey, Jackie. "Ingredients in Hand Sanitizer". LIVESTRONG.COM. Retrieved 2018-06-11.
^ Janusz Szajewski, MD, Warsaw Poison Control Centre (August 1991). "Propylene glycol (PIM 443)". IPCS INChem. Retrieved July 2, 2009.
^ Pucker AD, Ng SM, Nichols JJ (2016). "Over the counter (OTC) artificial tear drops for dry eye syndrome". Cochrane Database Syst Rev. 2: CD009729. doi:10.1002/14651858.CD009729.pub2. PMC 5045033. PMID 26905373.
^ "What's That Stuff? Aircraft Deicers". Chemical & Engineering News. American Chemical Society. 2000-07-10. Retrieved 2013-06-21.
^ "5 Ways to Winterize a Vacant Home". wikiHow. 2012-06-11. Retrieved 2014-02-27.
^ "Properties of Some Particular Solutions" (PDF). Portal del DMT. Retrieved 2014-02-27.
^ Salnick, Robert (2010-08-04). "Windborne in Puget Sound: Why does a holding plate work?". Windborneinpugetsound.blogspot.com. Retrieved 2014-02-27.
^ "Material Safety Data Sheet: Winter Care RV Antifreeze" (PDF). Chemical Specialties. Retrieved 2014-02-27.
^ Nielsen, Nicolaj. "Propylene glycol for dairy cows". Animal Feed Science and Technology. 115: 191–213. doi:10.1016/j.anifeedsci.2004.03.008.
^ Hamilton, D. J. (1890). "Gastric Dyspepsia". The Lancet. 2: 306. doi:10.1016/S0140-6736(02)17110-8.
^ "Material Safety Data Sheet Propionaldehyde MSDS". ScienceLab.com. 2010.
^ Miller DN, Bazzano G; Bazzano (1965). "Propanediol metabolism and its relation to lactic acid metabolism". Ann NY Acad Sci. 119 (3): 957–973. Bibcode:1965NYASA.119..957M. doi:10.1111/j.1749-6632.1965.tb47455.x. PMID 4285478.
^ Ruddick JA (1972). "Toxicology, metabolism, and biochemistry of 1,2-propanediol". Toxicol Appl Pharmacol. 21: 102–111. doi:10.1016/0041-008X(72)90032-4.
^ rocklinusd. "Leathal dose table" (PDF).
^ Alton E. Martin - Frank H. Murphy DOW CHEMICAL COMPANY. "GLYCOLS - PROPYLENE GLYCOLS" (PDF).
^ Flanagan RJ; Braithwaite RA; Brown SS; Widdop B; de Wolff FA. The International Programme on Chemical Safety: Basic Analytical Toxicology. WHO, 1995.
^ ab U.S. Food and Drug Administration (FDA). "Subchapter B - Food for Human Consumption. § 184.1666. Propylene glycol." Code of Federal Regulations, 21 C.F.R. 184.1666
^ National Library of Medicine;.Propylene glycol is used in antifreezes Human Toxicity Excerpts: CAS Registry Number: 57-55-6 (1,2-Propylene Glycol). Selected toxicity information from HSDB. 2005.
^ Gaunt, I. F.; Carpanini, F. M. B.; Grasso, P.; Lansdown, A. B. G. (1972). "Long-term toxicity of propylene glycol in rats". Food and Cosmetics Toxicology. 10 (2): 151–162. doi:10.1016/S0015-6264(72)80193-7.
^ "Database of Select Committee on GRAS Substances (SCOGS) Reviews". FDA. Retrieved 2016-05-11.
^ FDA. "Subchapter B - Food for Human Consumption. § 182.1. Substances that are generally recognized as safe." Code of Federal Regulations, 21 C.F.R. 182.1
^ Agency for Toxic Substances and Disease Registry (2008). "Addendum to the Toxicological Profile for Propylene Glycol": 7.
^ Robertson, OH; Loosli, CG; Puck, TT; Wise, H; Lemon, HM; Lester, W (September 1947). "Tests for the chronic toxicity of propylexe glycol and triethylene glycol on monkeys and rats by vapor inhalation and oral administration". JPET. 91 (1): 52–76. PMID 20265820.air containing these vapors in amounts up to the saturation point is completely harmless
^ "DOW: Product Safety Assessment (PSA): Propylene Glycol". Retrieved 2011-11-09.
^ A Guide to Glycols, Dow, page 36
^ "Everyday Substances Increase Risk of Allergies in Children, Swedish Study Reveals". ScienceDaily. Oct 19, 2010.
^ "Chemical Compounds Emitted From Common Household Paints and Cleaners Increase Risks of Asthma and Allergies in Children". Harvard. Retrieved 3 October 2018.
^ Choi, Hyunok; Norbert Schmidbauer; Jan Sundell; Mikael Hasselgren; John Spengler; Carl-Gustaf Bornehag (2010-10-18). Hartl, Dominik, ed. "Common Household Chemicals and the Allergy Risks in Pre-School Age Children". PLoS ONE. 5 (10): e13423. Bibcode:2010PLoSO...513423C. doi:10.1371/journal.pone.0013423. PMC 2956675. PMID 20976153.
^ 1,2-Dihydroxypropane SIDS Initial Assessment Profile <"Archived copy" (PDF). Archived from the original (PDF) on 2009-02-19. Retrieved 2008-01-08.CS1 maint: Archived copy as title (link) >, UNEP Publications, SIAM 11, U.S.A, January 23–26, 2001, page 21.
^ Title 21, U.S. Code of Federal Regulations. 1999.
^ Szajewski, Janusz. "Propylene Glycol (PIM 443)." 1991. 2 June 2010 http://www.inchem.org/documents/pims/chemical/pim443.htm#SectionTitle:9.1%20%20Acute%20poisoning
^ Speth, P. A. J.; Vree, T. B.; Neilen, N. F. M.; De Mulder, P. H. M.; Newell, D. R.; Gore, M. E.; De Pauw, B. E. (1987). "Propylene Glycol Pharmacokinetics and Effects after Intravenous Infusion in Humans". Therapeutic Drug Monitoring. 9 (3): 255–258. doi:10.1097/00007691-198709000-00001. PMID 3672566.
^ Seidenfeld, M. A.; Hanzlik, P. J. (1932). "The general properties, actions, and toxicity of propylene glycol". J Pharmacol Exp Ther. 44: 109–121.
^ Demey, H.; Daelemans, R.; De Broe, M.E.; Bossaert, L. (1984). "Propylene glycol intoxication due to intravenous nitroglycerin". The Lancet. 323 (8390): 1360. doi:10.1016/S0140-6736(84)91860-9. ISSN 0140-6736.
^ FDA. "Subchapter E - Animal Drugs, Feeds, and Related Products; § 582.1666. Propylene glycol." Code of Federal Regulations, 21 C.F.R. 582.1666
^ Peterson, Michael; Talcott, Patricia A. (2006). Small animal toxicology. St. Louis: Saunders Elsevier. p. 997. ISBN 0-7216-0639-3.
^ "pg_cats_latest.PDF" (PDF). Retrieved 2013-06-21.
^ American Medical Association, Council on Drugs (1994). AMA Drug Evaluations Annual 1994. Chicago, Illinois: American Medical Association: 1224. Missing or empty|title=(help)
^ ab Environmental Impact and Benefit Assessment for the Final Effluent Limitation Guidelines and Standards for the Airport Deicing Category (Report). Washington, D.C.: U.S. Environmental Protection Agency (EPA). April 2012. EPA-821-R-12-003.
External links
Propylene glycol website- WebBook page for C3H8O2
ATSDR - Case Studies in Environmental Medicine: Ethylene Glycol and Propylene Glycol Toxicity U.S. Department of Health and Human Services (public domain)- Propylene Glycol - chemical product info: properties, production, applications.
- ChemSub Online: Propylene glycol