Eschweiler–Clarke reaction
| Eschweiler–Clarke reaction | |
|---|---|
| Named after | Wilhelm Eschweiler Hans Thacher Clarke |
| Reaction type | Substitution reaction |
| Identifiers | |
| Organic Chemistry Portal | eschweiler-clarke-reaction |
RSC ontology ID | RXNO:0000376 |
The Eschweiler–Clarke reaction (also called the Eschweiler–Clarke methylation) is a chemical reaction whereby a primary (or secondary) amine is methylated using excess formic acid and formaldehyde.[1][2][3][4]Reductive amination reactions such as this one will not produce quaternary ammonium salts, but instead will stop at the tertiary amine stage. It is named for the German chemist Wilhelm Eschweiler (1860–1936) and the British chemist Hans Thacher Clarke (1887-1972).
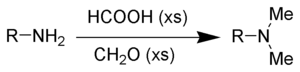
Mechanism
The first methylation of the amine begins with imine formation with formaldehyde. The formic acid acts as a source of hydride and reduces the imine to a secondary amine. The driving force is the formation of the gas carbon dioxide. Formation of the tertiary amine is similar, but slower due to the difficulties in iminium ion formation.

From this mechanism it is clear that a quaternary ammonium salt will never form, because it is impossible for a tertiary amine to form another imine or iminium ion.
Chiral amines typically do not racemize under these conditions.[5]
Altered versions of this reaction replace formic acid with sodium cyanoborohydride.
See also
- Leuckart–Wallach reaction
References
^ Eschweiler, W. (1905). "Ersatz von an Stickstoff gebundenen Wasserstoffatomen durch die Methylgruppe mit Hülfe von Formaldehyd". Ber. 38: 880. doi:10.1002/cber.190503801154..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ Clarke, H. T.; Gillespie, H. B.; Weisshaus, S. Z. (1933). "The Action of Formaldehyde on Amines and Amino Acids". Journal of the American Chemical Society. 55 (11): 4571. doi:10.1021/ja01338a041.
^ Moore, M. L. (1949). "The Leuckart Reaction". Org. React. 5: 301–330. doi:10.1002/0471264180.or005.07.
^ Icke, R. N.; Wisegarver, B. B.; Alles, G. A. (1945). "β-Phenylethyldimethylamine". Organic Syntheses.; Collective Volume, 3, p. 723
^ Farkas, Eugene; Sunman, Cheryl J. (1985). "Chiral synthesis of doxpicomine". J. Org. Chem. 50 (7): 1110. doi:10.1021/jo00207a037.)